AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog

Sodium chloride is an ionic compound composed of sodium cations, Na +, and chloride anions, Cl −, combined in a 1:1 ratio. Keep in mind, however, that the formula for an ionic compound does not represent the composition of a discrete molecule, so it may not correctly be referred to as the “molecular mass.”Īs an example, consider sodium chloride, NaCl, the chemical name for common table salt. The formula mass for an ionic compound is calculated in the same way as the formula mass for covalent compounds: by summing the average atomic masses of all the atoms in the compound’s formula. 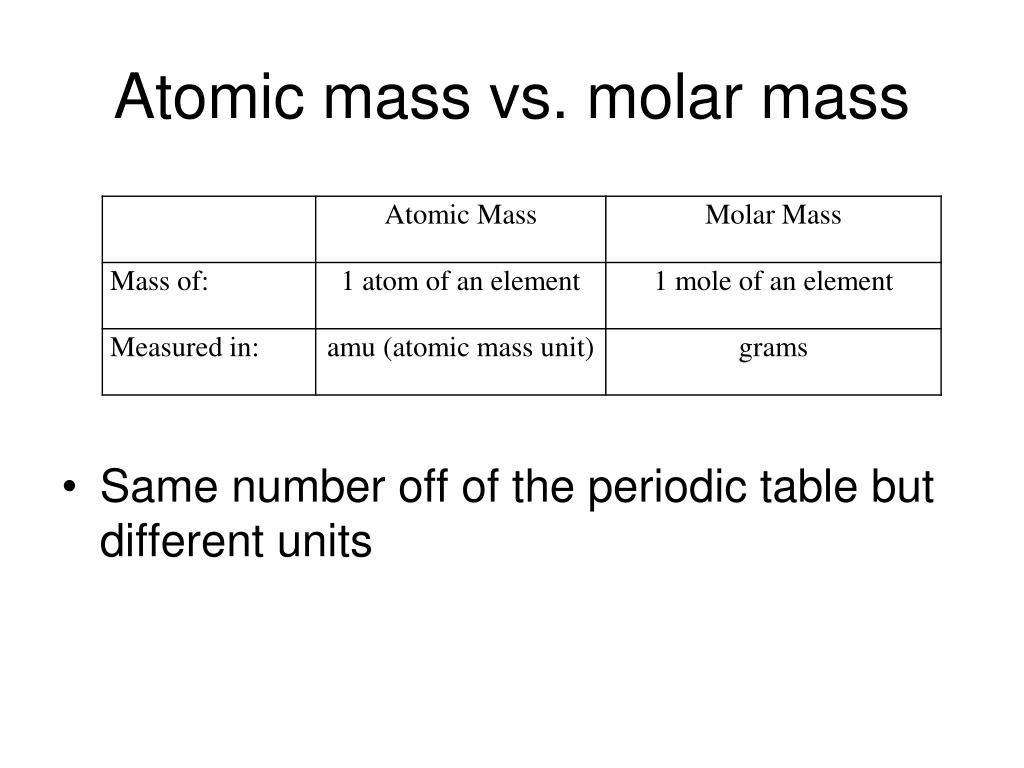
Ionic compounds are composed of discrete cations and anions combined in ratios to yield electrically neutral bulk matter. A black sphere, attached to two red spheres and a white sphere is attached to the black sphere on the top right of the six-sided ring. Attached to the first black sphere of that row is another red sphere. Attached to the last black sphere of that row are two more white spheres. Attached to the farthest right black sphere is a red sphere, connected to two more black spheres, all in a row. Attached to each of the four black spheres is one smaller white sphere. Six black spheres are located in a six-sided ring and connected by alternating double and single black bonds. The merged cell under the first five columns reads “Molecular mass.” To the left of the table is a diagram of a molecule. The fifth column contains the symbol “=” in each cell except for the last, merged cell. The fourth column contains the numbers “12.01,” “1.008,” and “16.00” as well as the merged cell. The third column contains the multiplication symbol in each cell except for the last, merged cell. The second column contains the numbers “9,” “8,” and “4” as well as the merged, cell. The merged cell runs the length of the first five columns. The header row reads: “Element,” “Quantity,” a blank space, “Average atomic mass (a m u),” a blank space, and “Subtotal (a m u).” The first column contains the symbols “C,” “H,” “O,” and a merged cell. The table is made up of six columns and five rows. Using the masses on the table provided, how many significant figures should your answer have had This problem has been solved Youll get a detailed solution from a subject matter expert that helps you learn core concepts. The model shows the molecular structure of aspirin, C 9H 8O 4. Use the periodic table below to calculate the molar mass of citric acid (C6H8O7). Molecular mass the sum of the atomic masses of each atom in the molecule. You will need to refer to a periodic table for proton values.\): The average mass of an aspirin molecule is 180.15 amu. In this notation, the atomic number is not included. Symbol-mass format for the above atom would be written as Cr-52. 
With aluminium, antimony and copper it forms highly magnetic alloys. Drinks cans are made of an alloy of aluminium with 1.5 manganese, to improve resistance to corrosion. This is extremely strong and is used for railway tracks, safes, rifle barrels and prison bars. For an example of this notation, look to the chromium atom shown below:Īnother way to refer to a specific atom is to write the mass number of the atom after the name, separated by a hyphen. Manganese steel contains about 13 manganese. The "A" value is written as a superscript while the "Z" value is written as a subscript. Both the atomic number and mass are written to the left of the chemical symbol. The composition of any atom can be illustrated with a shorthand notation called A/Z format.
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
